Embryo Selection: Toward a healthier society
Improving intelligence & defeating disease...
Written by Diana Fleischman, Ives Parr, and Laurent Tellier.
Parents want their children to have the best lives possible. The defining feature of parental love is a willingness to sacrifice to give one’s children the best chance of success, happiness, physical health, and emotional well-being.

Each year, millions of people use in vitro fertilization (IVF) to try to have children. IVF is a procedure in which women inject hormones to produce several eggs instead of the single egg they would usually produce in a normal ovulatory cycle. Eggs are retrieved and then mixed with sperm outside of her body (they are fertilized in vitro). The resulting embryo(s) grow in an incubator for a few days. Embryos are then typically tested, and (usually) one is chosen to be implanted while the rest of the embryos remain frozen for later use.
People use IVF because they are experiencing infertility, because they are in same-sex relationships, or because they want to prevent the transmission of heritable diseases. Most of the time, IVF cycles produce more than one embryo, and families choose which embryo to implant. This choice is consequential because even though full siblings share half of their genes, they can be radically different, as anyone with a sibling can attest.
This is where a new technology comes in: preimplantation genetic testing for polygenic disorders (PGT-P) or polygenic screening, which may inform which embryo parents choose and who is born. Because embryo choice is so consequential, polygenic screening—like other, new reproductive technologies before it—attracts more than its share of controversy and critics, many of whom use the label of eugenics as a smear, to suggest that parents electing to screen their own embryos are somehow akin to Nazis endorsing sterilization and murder. Progressives who criticize polygenic screening tend to use accusations of eugenics inconsistently, applying it to reproductive technology like polygenic screening but not abortion.
Other similar reproductive technologies are less controversial today than when first introduced. For example, most couples using IVF choose to genetically test their embryos for an abnormal number of chromosomes, known as “aneuploidy.” One reason for this genetic screening is that aneuploid embryos almost always result in a miscarriage. Another reason is that the few cases that do not miscarry result in life-long, incurable syndromes, such as Down Syndrome—associated with health problems, disability, and shorter lifespan. While aneuploidy screening is not entirely uncontroversial, this screen is far more widely accepted than when it was first introduced decades ago, and criticism of screening out Down Syndrome as “eugenics” is increasingly a fringe position.
Another commonly accepted embryo screen is for monogenic disorders, that is for diseases caused by single genes. For example, couples can screen out embryos with Tay-Sachs, a devastating neurological disorder that kills young children. Carriers of Huntington’s disease—a degenerative neurological disease that, on average, kills its victims in their early sixties - also often choose to use IVF to screen out embryos likely to be afflicted. For most people, it is clear today why future parents would not want their children to have chromosomal abnormalities or monogenic disorders. But the acceptance of these tests has taken time and was far from universal when these tests were introduced in the 1990s.
Many uses of monogenic screening—even those which today are among the most common and accepted—were initially hotly criticized, and not even approved for use in the country of their invention. Health authorities in the UK finally approved monogenic embryo screening against BRCA1 to reduce the risk of breast cancer only in the mid-2000s. At the time (2006), this ruling to allow BRCA1 screening in the UK was harshly criticized:
...the Human Fertilisation and Embryology Authority (HFEA), in approving this request, crossed a bright line separating legitimate medical genetics from the quest for ‘the perfect baby,’
Today in 2023, it is a crankish position to say that reducing breast cancer risk is crossing a “bright line” into illegitimate medical genetics and eugenics.
Polygenic screening and its discontents
But monogenic and chromosomal screening can only address a part of disease risk because most health conditions that afflict people are polygenic, meaning they are not simply caused by one gene or by a chromosomal abnormality. Instead, they are caused by a huge number of small additive effects dispersed throughout the genome. For example, cancer, schizophrenia, and diabetes can be best predicted by models using tens of thousands of genes.
A polygenic risk score (PRS) looks at a person’s DNA to see how many variants they have associated with a particular disease. Like BRCA1, polygenic risk scores are typically not determinative: “Polygenic screening is not a diagnosis: It is a prediction of relative future risk compared to other people.” In other words, someone with BRCA1 has a higher risk than someone without, and someone with a high breast cancer PRS has a higher risk than someone with a lower breast cancer PRS. But in principle, BRCA1 is just one gene out of thousands contributing to a PRS, with each bit contributing a small part of a total risk estimate.
But good health isn’t the only way that parents may try to ensure that their children have the best lives possible. Polygenic scores can be used to prioritize embryos predicted to have characteristics associated with happiness, success, and attractiveness, including height and intelligence. Polygenic scores cannot predict traits with perfect accuracy, but they can be used to choose the embryo most likely to be the tallest or longest-lived in a given embryo batch.
Many of the arguments against polygenic screening are similar to arguments made decades ago against IVF and other forms of embryo testing. Some have suggested that polygenic screening should not be available to couples until we have seen the outcomes of polygenic screening in adulthood, even though polygenic screening just changes the likelihood that one embryo will be chosen over another. IVF, which completely changes the way babies are conceived, and is therefore much riskier than polygenic screening in anticipation, was not held to this standard. Imagine that it had been. The first IVF baby, Louise Brown, is only 44 years old. If we had waited to see if she and others like her had developed any unforeseen health problems in adulthood more than 8 million babies would never have been born.
Polygenic screening has strong supporters and ardent critics. Julian Savulescu, who coined the term Procreative Beneficence thinks “polygenic embryo screening will revolutionize medicine and human enhancement." In contrast, behavioral geneticist Eric Turkheimer says it won’t work “well enough,” and people will lose interest (though current polling indicates people are very interested). Irish neurogeneticist Kevin Mitchell has called polygenic embryo screening “consumer eugenics,” and though he has not called for a ban, he has expressed concern with a rush to application. Ewan Birney, Director of the European Bioinformatics Institute, thinks that there is a "moral question" of "should we allow parents this control of their child's genetic makeup" (he believes the answer is no). Compared to Europe, where experts such as Birney show a keen desire to regulate such technology, there seems to be very little desire for regulation in the USA. During a debate at the most recent American Society for Reproductive Medicine meeting, all the experts, for and against, agreed that polygenic screening technology should not be regulated by the government. Dr. Lee Shulman, a critic of the technology, said “to make it illegal or to stop the development of [polygenic embryo screening] ultimately harms everyone.”
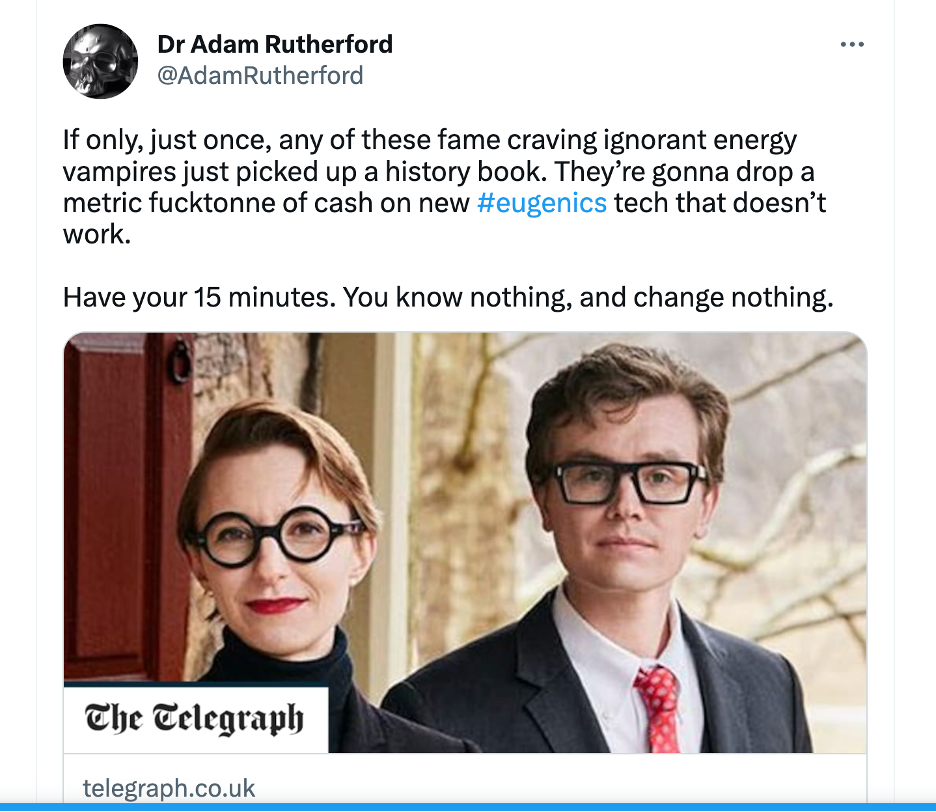
At the most extreme end, few critics have been as strident as Adam Rutherford. In his 2022 book Control: The Dark History and Troubling Present of Eugenics, Rutherford discusses eugenics’ “troubling present”, including several pages criticizing polygenic screening and its proponents. Here, Rutherford calls polygenic embryo screening “barely viable.” Rutherford calls proponents of PGT-P like Prof. Stephen Hsu “ill-informed” and “shallow-thinking provocateurs” (pg 198) comparing them to arsonists “who set fire to things and then walk away”. He characterizes people who “ponder…embryo selection for things like intelligence” as “careless” people who “let others clean up the mess they make” (pg 236).
Rutherford further writes that he cannot see how embryo selection for “intelligence, or to curtail the risk of mental problems” will result in a “plausible—or desirable--future” (pg 237). Recently he tweeted that Simone and Malcolm Collins, who used Genomic Prediction’s polygenic health score to choose an embryo with the lowest risk of common health problems (Titan, pictured above), are “fame craving ignorant energy vampires”.
Is polygenic screening eugenics? Is it immoral?
In Control, Rutherford argues that advocating any policy that could be considered eugenics is a slippery slope to unethical government control, like forced sterilization. Given this position, we should expect consistency in how eugenics is defined. But Rutherford has taken various positions on whether embryo screening is eugenics. Within one year, Rutherford has said:
When criticizing IVF patients on Twitter, embryo selection is “new eugenics tech that doesn’t work”.
When promoting his book in an interview, embryo selection could be eugenics, but it doesn’t really matter, and “I shy away from calling those [IVF embryo screening] interventions eugenics”
In Control: “Whether these qualify as eugenic interventions is up for debate, but in some ways I don’t really care.” (pg 213)
On a podcast: “Giving individuals autonomy, granting them control over how they reproduce is powerfully anti-eugenic" echoing Paige Harden in an interview for the BBC.
Is polygenic screening eugenics? Since it was invented, preimplantation genetic testing has been defended by its Nobel prize-winning practitioners against accusations of “eugenics.” Some draw a line defining coercive state-sponsored programs (like forced sterilization) as “eugenics,” while programs of free parental choice—such as parents choosing whether to abort a fetus with Down Syndrome—as “not eugenics.” But this may be a false dichotomy. Most people and many ethicists endorse policies that can be characterized as eugenics, like laws prohibiting brothers and sisters from marrying, or routine testing for fetal abnormalities.
The “eugenics” label seems to do little to enlighten us about ethics. However, we reject the framing of embryo selection for traits or diseases as a frightening form of technology that raises the same moral problems as forced sterilization programs or gas chambers. Polygenic screening must be acknowledged to be in the domain of free parental choice and reproductive liberty, regardless of whether this parental choice results in a preference for health, height, or happiness.
What Rutherford gets wrong about embryo screening:
Those who consider themselves on the right side of history often do not feel compelled to make the best evidence-based arguments. Rutherford, in particular, spends several pages of Control using guilt-by-association tactics to malign Steve Hsu and only one paragraph on antagonistic pleiotropy (tackled below). Since Rutherford is so outspoken against polygenic embryo screening, and because he endorses these views on a large public platform, we dug into his arguments in Control and elsewhere, summarizing them— along with our rebuttals—below:
Control focuses little on terrible diseases that embryo screening might prevent
In Control, Rutherford makes a point we all agree with:
“Diseases exist, and thousands have genetic causes, and we must treat, cure and, if possible, eradicate them. Any other position is morally indefensible.” (pp. 153-154)
Rutherford’s critique of polygenic embryo screening focuses almost entirely on intelligence and height and barely mentions that polygenic screening can be used to select against embryos with diseases like Alzheimer’s, coronary artery disease, heart attack, hypertension, schizophrenia, diabetes, and cancer. This omission of the most commonly practiced and effective use of polygenic screening is a necessary rhetorical strategy for Rutherford. It would be nearly impossible to both argue the humane position that heritable diseases should be eradicated and that polygenic screening is “careless” (pg 236) and “barely viable” (pg 211).
For example, Titan Collins, pictured above, was selected on the basis of this polygenic health index, made up of 20 disease risks, but not IQ or height. Her genome was particularly chosen for a low risk of the cancer that took her grandmother’s life before her birth. Here is a clip of Simone Collins showing how she chose Titan from an embryo ranking for health risk.
Using data from the UK BioBank, Widen et al. modeled that an embryo genetically selected using this health index would live an average of 3-4 disability-adjusted years longer than an embryo selected by chance, when genetically selecting the healthiest from among 10 embryos, in a couple with European ancestry (such as the Collins). The same paper showed that, had the Collins couple been of a different ancestry, this gain would have been half this number of years—significantly less, but also significantly better than selecting the embryo by chance. These peer-reviewed findings were validated empirically in tens of thousands of sibling pairs, and—where similar work was attempted by other researchers—validated with similar rates of disease incidence reduction from embryo selection (Turley et al, 2021 and Lencz et al. 2021).
Some have contended that these validations do not prove the case because they are based on population models rather than clinical outcomes. This is similar to the objection to BRCA1 screening in 2006. Now BRCA1 mutation screening is routinely practiced after decades of debating that it is “not a sure thing.” With polygenic screening, adult siblings have the same relationship with one another as embryos sequenced in vitro. Polygenic scores reasonably predict characteristics like height or health in siblings, it logically follows they have predictive power in embryos. This argument against using probabilistic predictors was misguided when it was used against BRCA embryo screening in 2006, and it’s misguided when used against polygenic embryo screening now.
Polygenic screening can also reduce incidence of a debilitating mental illness—schizophrenia—more than any known environmental intervention. Rutherford does mention that schizophrenia is polygenic, and he cites papers from Shai Carmi, so his failure to acknowledge that Carmi arrives at the same conclusions about embryo screening for schizophrenia risk as Genomic Prediction is, again, omitted because it’s inconvenient to Rutherford’s argument. Carmi’s lab found that even when selecting among just two embryos, selecting the embryo with the lowest risk for schizophrenia, for example, may reduce disease risk by 34%, and that reduction of disease risk by 50% is possible when selecting from among 5 embryos. These are the same numbers found by Genomic Prediction—a very close and fully independent replication of their scientific result by a completely disinterested third party.
50% may not be a big deal for a person with average schizophrenia risk, but for people with a family history of schizophrenia, such technology is life-changing. Laurent Tellier, a co-founder of Genomic Prediction, helped a patient who decided to have children because polygenic embryo screening gave her an opportunity to reduce the risk of passing on the schizophrenia diagnosis suffered by her mother—a risk which is more than 10x elevated in persons who have a first-degree relative diagnosed with the disorder.
In contrast, Rutherford says that he is in favor of disease screening for a woman who is already pregnant—which is to say, abortion of a fetus found to be at high risk for a given disorder. He says: “the decision to terminate a pregnancy because of a prenatal diagnosis is something that I believe is an absolute personal choice and should be an unstigmatized right for women and parents. To do so is not eugenics”. (pg 181). Why is it, then, that a woman choosing to have an abortion is not eugenics, while an IVF patient like Simone Collins, selecting an embryo with the lowest risk of a disease, is publicly attacked by Rutherford on Twitter as a eugenicist and accused of being unattractive for good measure?
Rutherford mistakenly claims that polygenic screening is “barely viable”
Rutherford says:
A team of American and Israeli scientists modeled the possible utility of embryo selection in 2019, as proposed by people like Stephen Hsu. The results are not very impressive. The researchers used a battery of statistical techniques to work out what range of height or intelligence might emerge during a hypothetical round of IVF. The answers came out at just under an inch, and 2.5 IQ points. The typical error margins around IQ are about plus or minus 5 points, meaning that when you get an IQ score of say 120, it was actually somewhere between 115 and 125 when you took the test. And as for height, well, I can get up to six feet tall with the right shoes and hair product. So, from a practical point of view, embryonic selection for complex polygenic traits is barely viable. It’s a costly, technical procedure for arguably marginal gains that could be applied to a handful of people already undergoing IVF. (pg 211)
Even though Control doesn’t give polygenic screening for diseases a fair hearing, Rutherford does distinguish between traits, such as height and intelligence, from diseases. So, is trait screening barely viable? Here Rutherford cites this 2019 study from Karavani et al, but he fails to mention an important aspect of this study. According to the paper, the top-scoring embryo is expected to be 2.5cm or 2.5 IQ points above the average embryo. 2.5 IQ points does not represent the entire range, which can be 5 points or higher. This standard of comparing the benefit of testing relative to an average embryo isn’t the standard people commonly use with other pregnancy-related outcomes. For example, no one would advise a woman not to have prenatal genetic testing to avoid rare outcomes just because her “average” pregnancy is likely to be genetically normal.
That Rutherford chose to include the low estimate from a paper that gives a range of estimates concerning the predictive power of models is telling. Yet, as the authors make clear, even if we use the most conservative estimates from 2019, the science in this area is moving rapidly, and the best models continue to predict larger variance between embryos. Rutherford’s argument is already out of date.
The point of selection between embryos is to increase the chance that a child will have a lower disease burden and more desirable positive outcomes. This is already how parents make decisions. A mother might not be guaranteed that her child will reach a certain lifespan or height or IQ or level of happiness if she feeds him nutritious food, but she will nonetheless do the best she can, often with far more modest evidence for efficacy than in the case of cutting-edge genetic selection. There is no evidence that parents using polygenic screening imagine that they are guaranteed a certain outcome, but given a choice between embryos, they still want to prioritize a healthier embryo than they would by chance. This question has been studied in real patient cohorts of parents using polygenic embryo screening, showing that 90% of patients given this information elect to use it to make their choice.
Rutherford underestimates the comparative utility of genetic selection
Rutherford presents environmental interventions as more effective for improving outcomes than polygenic screening and simultaneously presents polygenic screening as if it will somehow prevent or discourage improving environmental interventions. There is no evidence to support either contention in the developed world.
Rutherford says:
We know how to raise the IQ of the population already — you do it by improving childcare and reading books; improving teaching practices; providing better access to sanitation, medical care and sports facilities.
“Medical care” and “sanitation” might be associated with increases in intelligence if you consider providing clean water, deworming, or supplementing iodine for children who are severely iodine deficient—none of which are relevant to the developed world. There is no evidence that access to sports facilities improves IQ.
Previously, Rutherford stated that an average gain of 2.5 IQ points isn’t impressive. But the one thing that does show substantial improvements in IQ in the developed world is reducing lead exposure. Since 2015, the average loss in cognitive ability in the USA due to lead is 2.6 IQ points—the same as the impact on IQ from embryo screening estimated in 2019. In our view, the benefit from removing exposure to lead poisoning doesn’t return “marginal” gains any more than polygenic embryo screening does. And there is no reason to believe that parents who choose to reduce lead exposure to their child will therefore be any less likely to care for their children’s education or to provide their children with books.
Rutherford focuses on interventions that can be “deployed to populations''. However, polygenic screening, so far, is administered to individuals. It doesn’t make any sense to pit potential interventions of the welfare state or greater redistribution against polygenic screening because the wealthy (by global standards) who choose to pay for this technology wouldn’t otherwise be donating money to iodine supplementation or deworming.
Rutherford makes IVF sound rare, traumatic, and expensive
Rutherford says:
IVF is not exactly an easy procedure. Any woman who has been through it will confirm this readily. It’s a lot less fun than sex, and people who go through this arduous, emotionally fraught and often physically painful process mostly do it as a last resort to have kids when the standard options have not borne fruit. It’s not fun and it’s not a game. You may have noted that most of the people who knock around the idea of embryo selection tend not to be the ones who will have to endure the daily rounds of injections to induce ovarian hyperstimulation, or the needle through the vaginal wall to get access to the ovarian follicle. The people who seem most excited by the idea of eliciting molecular control over reproduction don’t tend to have wombs at all (pg 211).
Rutherford posits that since women are underrepresented publicly advocating for polygenic screening, they are therefore less in favor of it than men. The other suggestion is that being in favor of polygenic screening will somehow impose suffering on women that they wouldn’t choose themselves. Rutherford is essentially saying that the anti-polygenic screening position is a feminist position, similar to being against commercial surrogacy or sex work—a woman might be pushed into polygenic screening by eugenic patriarchy and thus disproportionately harmed.
Rutherford presents no data about women’s experiences with IVF. But I, Diana, have been through the most intense part of IVF, ovarian stimulation, and egg retrieval as an altruistic egg donor five times. My experiences with IVF were mildly uncomfortable and in no way traumatic—so much so that I was willing to go through IVF multiple times essentially for free. Also, Rutherford seems to forget that, for most women, a successful IVF process culminates in pregnancy and birth. To me, the discomfort of IVF is on par with a month of second-trimester pregnancy. I would much rather go through IVF than endure even three hours of early labor!
Simone Collins, whom Rutherford vilified on Twitter, has been through IVF six times in order to get pregnant. After five rounds she had more than enough embryos, but did the sixth round to utilize polygenic screening. Simone says
IVF is empowering. Initially it seemed intimidating and IVF is stigmatized and presented as stressful and painful, which makes it seem that much more onerous.
Both Simone and I also didn’t have any difficulties recovering within a few hours from egg retrieval. Regarding Rutherford’s claims about IVF, Simone reacted:
[…] as if nobody has ever referred to trying to conceive a baby through sex as an ‘arduous, emotionally fraught and often physically painful process!’
To women like me and Simone, Rutherford’s benevolent concern is a fig leaf over condescending paternalism.
IVF is also not nearly as rare or expensive as Control suggests (e.g. “handful of people already undergoing IVF”). For example, in 2022, 11.5% of babies in Denmark were born with the assistance of reproductive technology (ART) including IVF. In 2021 in the USA, more than 89,000 babies, or more than 2% of live births in the USA, were the result of ART and there were more than 24,000 egg freezing cycles. Each year roughly 2 million IVF embryos are genetically screened worldwide. Paying for IVF privately is expensive, as much as $20,000 a cycle. But IVF is sometimes covered by insurance, and IVF is increasingly covered by governments hoping to increase the birth rate. It’s subsidized in Denmark, Israel, and soon—and most consequentially—it will be subsidized in China. And given the pro-embryo screening attitudes in China it’s possible that many of these subsidized IVF cycles will also utilize polygenic screening.
The cost of polygenic screening is small compared to the cost of many other child-related expenses. For example, Genomic Prediction screens embryos for $1,000 to start and then $400 for each embryo. Screening an average IVF cycle batch of five embryos costs $3,000, which is what many people pay for a few months of daycare or private school. For anyone doing IVF in the USA, 50% of IVF cycles are already spending ~$1500 on genetic testing anyway, so the added expense is even less. In the future, governments may decide that polygenic screening is a good investment. The cost to the state of treating the disabilities that screening could prevent and the value of human capital of healthier citizens are both much larger than the cost of subsidized embryo screening.
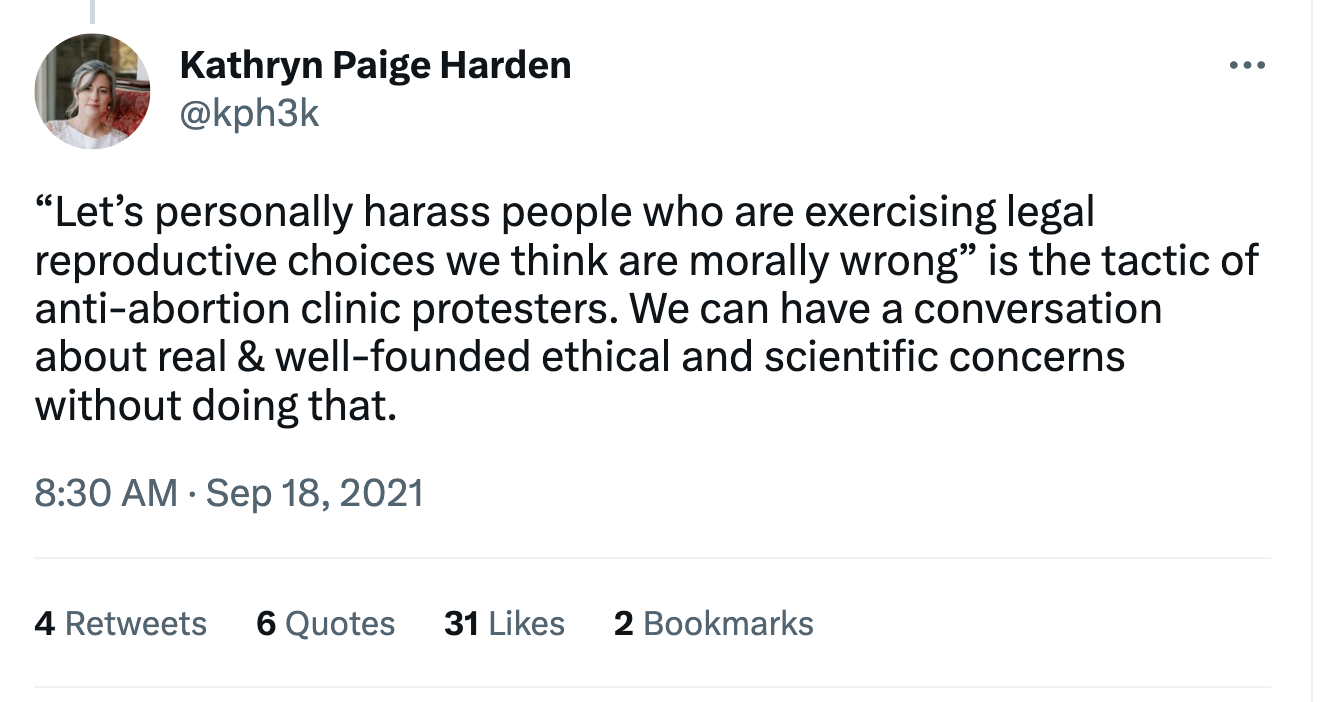
If polygenic screening really doesn’t work and is a waste of money, then mothers like Simone should not be disparaged by men like Adam Rutherford, at least not more than if she buys a luxury handbag. And if polygenic screening does work, or even if it might work, then the mothers who use it are demonstrating admirable maternal love that strives to ensure the health and wellbeing of their children. That’s why the public shaming of people who screen their embryos for health like Simone Collins and Rafal Smigrodzki is absurd. The discourse about Smigrodzki got so heated on Twitter that Paige Harden, a behavioral geneticist, felt compelled to tell fellow progressives that harassing people who are exercising their legal reproductive choices is the same tactic used by abortion clinic protesters.

Rutherford focuses on “unforeseen health problems” but not unforeseen health benefits
Rutherford says he wouldn’t want to select embryos because:
[…] we don’t know if selecting for something means you are inadvertently selecting against something else. One study found that IQ positively correlates with anorexia, anxiety disorders, attention deficit hyperactivity disorder and asthma…a nudge taller or smarter, you may also be nudging that child toward an eating disorder or other unforeseen health problems (pg 211).
Here Rutherford is alluding to antagonistic pleiotropy. This occurs when genes that are beneficial in one way are harmful in another. But the reality is that, more often, when we select against some diseases, we inadvertently reduce the risk of other diseases.
To make his case that IQ is associated with a number of bad outcomes, Rutherford cherrypicks a single study of Mensa members. Mensa is a society for people who score in the top 2% of IQ. Mensans are unlikely to be representative of high IQ people generally, and this Mensa study is contradicted by numerous other studies. There is a large body of evidence that smarter people tend to have fewer physical and mental health problems. Moreover, Widen et al. (2022) found that “correlations between disease risks are found to be mostly positive, and generally mild,” a finding which “supports the folk notion of a general factor which characterizes overall health”—which, in the paper, extends to the diseases which Rutherford is concerned with. The mild positive correlations suggest that selecting against one disease will, on average, lead to selection against other diseases, not for them.
There is evidence that directly contradicts Rutherford’s contention. Alleles associated with higher IQ are protective against schizophrenia, ADHD, and Alzheimer’s, and alleles associated with intelligence are associated with not being a smoker, being taller, being less likely to be depressed, being less neurotic, and having a lower body mass index. Sniekers et al. (2017) did not find a significant association between variants associated with intelligence and anorexia. The bulk of the evidence contradicts Rutherford’s alarmist speculation about the negative outcomes of selecting for intelligence.
Polygenic screening isn’t “careless”
In discussions of embryo selection, it must be reiterated that couples face the embryo choice problem: they must select an embryo if they want a child. Any criticism of polygenic screening must be a relative comparison between methods. After embryos are determined to be free from aneuploidy and monogenic disorder, couples have to pick from the remaining embryos. If they decide not to use polygenic screening, they can use another method like visual inspection or picking randomly. Rutherford characterizes polygenic screening as “careless” (pg 236). We can think of no other context in which a deliberate, informed choice is considered more careless than a random choice. Embryo selection by mere visual inspection or by randomly choosing an embryo is not “safer” or “more proven” to improve health than polygenic selection.
Regardless of how you have a child, whether through natural conception or through IVF with polygenic screening, there are choices with tradeoffs. A lesbian couple cannot know for sure if the tallest sperm donor or the one with the most prestigious education will give their kids the best chances in life. There are no randomized controlled trials that will tell a couple whether they should try for a baby now while they are younger, or later when they have more money for quality childcare. There are many choices we know lead to beneficial outcomes, on average with varying levels of uncertainty. Presenting polygenic screening as unusual in this regard is transparently ideological.
Conclusion
Many arguments against polygenic screening look similar to arguments made against IVF decades ago. They smack of medical paternalism, as do many of Rutherford’s arguments in Control. But informed choice and reproductive autonomy are some of the cornerstones of modern medical ethics. Both principles imply that people who wish to use polygenic embryo selection should be free to do so.
Recently, a group of European scientists argued that polygenic screening should not be available to couples because it will lead to stigmatization, exacerbate inequalities, or lead to confusion by parents about how to weigh up information about risks before they decide which embryo to implant. These are indeed challenges, but they are not unique to embryo selection using polygenic scores, and they are not plausible arguments for restricting the autonomy of parents who wish to screen their embryos for polygenic traits. Furthermore, from an ethical perspective, it is unconscionable to deny polygenic screening to families with a history of any disease whose risk can be reduced by this lifesaving technology.
Many new technologies are initially only available to people with more money, but these first adopters then end up subsidizing research that drives costs down and quality up. Many other medical choices involve complexity or might result in some people being stigmatized, but this is a reason to encourage genetic counseling and to encourage social tolerance. It is not a reason to marginalize, stigmatize, or criminalize IVF mothers and fathers who wish to use the best available science to increase the chances that their children will be healthy and happy.
Learn more about the authors:
Diana Fleischman, Ives Parrhesia, Laurent Tellier.




1) They don't want to admit that some people are better than others, inherently. Boo hoo.
2) You put a scorecard of embryos in front of everyone, and everyone has a pretty good ballpark estimate of which are better and which are worse. Nobody is going to pretend equality is true when they are choosing their kids genes.
3) So bad feels.
4) Must therefore retard all human progress and cause immense suffering because don't want to deal with bad feels.
That's the anti-polygenic argument in a nutshell. I don't expect it to be very effective. At best it will cause it to take a bit longer before poor people have access.
It’s all eugenics, bud. Embryo selection (for whatever reason). Abortion. Assisted suicide. Giving out hormonal birth control like candy to teenagers and the poor while making it harder with every economic policy we adopt for the average person to have the kids they want. It’s allllllll eugenics